|
We found that the dual tag, his(6)-tag-maltose-binding protein (MBP), intended to facilitate purification and enhance proteins' solubility, disrupted such a pipeline, requiring additional screening and purification steps. 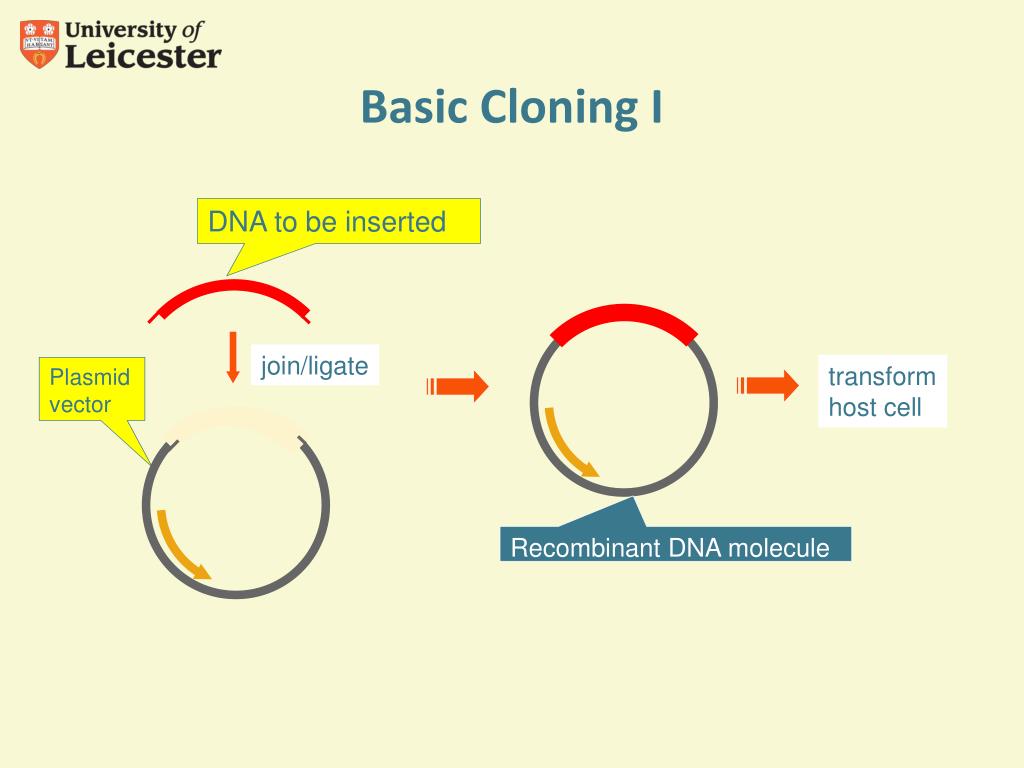
Production of milligram quantities of numerous proteins for structural and functional studies requires an efficient purification pipeline. The ScoAB complex was purified by standard high-throughput metal-ion affinity chromatography procedures, crystallized robotically, and its structure was determined by molecular replacement. Co-expression, in contrast, generated active transferase that catalyzed the predicted reaction. When expressed separately and mixed, the ScoAB subunits failed to interact productively no transferase activity was detected, and S-tagged ScoB failed to co-purify with His-tagged ScoA. Replacement of the His-tag encoding region of pMCSG7 with a sequence encoding the S-tag enabled selective purification of interacting proteins based more » on the His-tag associated with one member of the complex. Transfer of the LIC region of vector pMCSG7 into commercial vectors with alternative, compatible origins of replication allowed introduction of standard LIC PCR products into the vectors by uniform protocols. High-throughput, ligation-independent cloning (LIC) vectors used extensively for production and purification of single proteins were modified to allow simultaneous expression of interacting proteins and selective purification of functional complexes. The Bacillus subtilis genes scoA and scoB encode subunits of the heteromeric enzyme ScoAB, a putative succinyl-CoA:acetoacetate coenzyme A transferase. The engineered galectin-1 vector could prove to be a valuable tool for expressing other proteins in E. The vector also includes a pair of adjacent, unique restriction sites that will allow insertion of additional modules between the his-tag and the cleavage site of the more » leader sequence to generate a family of vectors suitable for high-throughput production of proteins. 
The protein was purified and cleaved under different conditions to simulate both plate-based screening methods and large-scale purifications for crystal production. Purification and cleavage of a target protein showed that the his-tag and the TEV cleavage site function properly. The vector functioned as expected, giving high cloning efficiencies and strong expression of proteins. The new vector incorporates an N-terminal his-tag followed by the TEV protease recognition site and a SspI restriction site used for LIC. This protease is highly specific and functions under a wide range of conditions. We have developed a new LIC vector, pMCSG7, that incorporates the tobacco etch virus (TEV) protease cleavage site into the leader sequence. Ligation-independent cloning (LIC) is well suited to robotic cloning and expression, but few LIC vectors are available commercially. Automated, plate-based methods for cloning, expression, and evaluation of target proteins will help researchers investigate the vast numbers of proteins now available from sequenced genomes. Somatostatin type 2 receptor antibody enhances mechanical hyperalgesia in the dorsal root ganglion neurons after sciatic nerve-pinch injury: Evidence of molecular and behavioral studies.To establish high-throughput methods for protein crystallography, all aspects of the production and analysis of protein crystals must be accelerated.Application of Engineered Bacteriophage T7 in the Detection of Bacteria in Food Matrices. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
